Biochips for better cancer therapy
One promising area of cancer treatment is photodynamic therapy, which combines the agents of a photosensitive drug, light, and oxygen.
Cancer is the second leading cause of disease-related death in the United States, and may overtake heart disease without aggressive new therapies. One promising area of cancer treatment is photodynamic therapy (PDT), which combines the agents of a photosensitive drug, light, and oxygen to attack cancerous tumors and lesions locally in the targeted region of the body by selective optical illumination.

 Enlarge
Enlarge
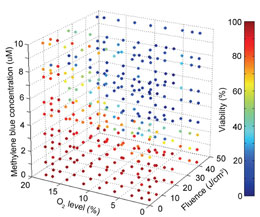
Research being conducted by Prof. Euisik Yoon’s group aims to dramatically accelerate progress in PDT. And it is being accomplished through a lab-on-a-chip measuring about the size of a quarter. At the heart of this biochip is a 5x5mm testing area that will test the interaction of the drug, light, and oxygen simultaneously, generating results in a fraction of the time of current testing practices.
“In cancer research doctors are always looking for better drugs,” explained Dr. Xia Lou, a postdoctoral fellow in Prof. Yoon’s group. “But there has always been a lack in the ability to efficiently test new drugs. Researchers are also hoping to get more reliable test results than is the norm. We are providing more precise drug test conditions to insure that the results we are getting will more closely match the actual results from cancer treatment.”
“PDT is a fairly complicated therapy because it requires light, oxygen and the drug, all three of which need to be carefully controlled,” said Dr. Lou. “With chemotherapy, for example, you only control how much of the drug is used.”
Photodynamic therapy uses a unique type of drug, called a photosensitizer, that generates high energy oxygen when activated by light, in this case an LED laser. The oxygen reaction destroys cells locally only in the immediate surrounding area, which in this case is the tumor, without damaging other healthy cells in the rest of the body. PDT may also prompt the patient’s immune system to attack the tumor, whereas without treatment it will likely ignore it.
 Enlarge
Enlarge
There are two primary challenges in PDT today. One is determining the best treatment plan for the patient, and the other is testing the efficacy of new and existing drugs. For example, the location of a tumor in the body will determine the amount of light needed to excite the drug, and will impact the amount of drug that is administered. Also, when testing new drugs, it is again important to know the amount of light needed to excite the oxygen reaction, and the strength of the reaction. This adds a high level of complexity compared to conventional drug testing that only tests one component, the drug itself.
Current PDT testing methods might take more than 24 hours to acquire just 10 data points. “With our device,” said Dr. Lou, “we can finish the testing in 1 hour and have 1,000 data points. The result is a very comprehensive understanding of how the 3 elements need to be manipulated for maximum effect in an individual patient.”

 Enlarge
Enlarge
The researchers reported the details of their findings in the February 2014 issue of Lab on a Chip, published by the Royal Society of Chemistry. The paper, “A high-throughput photodynamic therapy screening platform with on-chip control of multiple microenvironmental factors,” was authored by Dr. Xia Lou, Gwangseong Kim, Hyung Ki Yoon, Yong-Eun Koo Lee, Prof. Raoul Kopelman, and Prof. Euisik Yoon.
Directing the project are Prof. Euisik Yoon, Director of the Solid-State Electronics Laboratory and the Lurie Nanofabrication Center, and Prof. Raoul Kopelman, who is the Kasimir Fajans Collegiate Professor of Chemistry, Physics and Applied Physics, and a member of the Center for Biological Nanotechnology. Dr. Lou began working on the project as a doctoral student in Prof. Yoon’s group. The remaining investigators are members of Prof. Kopelman’s group.
This work was supported in part by the NSF Engineering Research Center for Wireless Integrated Microsystems (WIMS), Thermo Fisher Scientific, and the National Institute of Health.
 MENU
MENU 
